
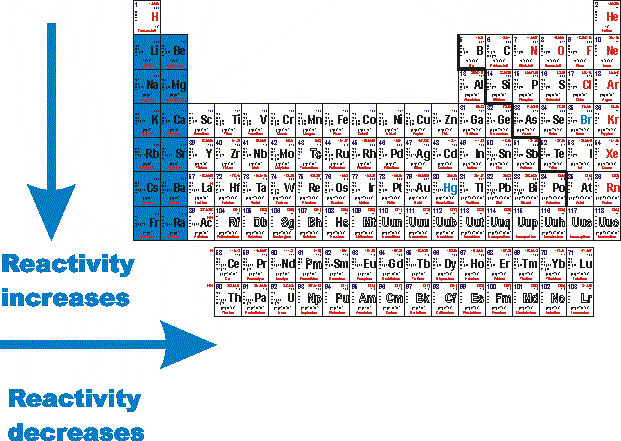
For example, it is commonly asserted that the reactivity of alkali metals ( Na, K, etc.) increases down the group in the periodic table, or that hydrogen's reactivity is evidenced by its reaction with oxygen. Both factors are actually distinct, and both commonly depend on temperature. It appears to embody both thermodynamic factors and kinetic factors (i.e., whether or not a substance reacts, and how fast it reacts). Reactivity is a somewhat vague concept in chemistry. The term reactivity is related to the concepts of chemical stability and chemical compatibility. equilibrium point of the reaction (i.e., the extent to which all of it reacts), and.variety of substances with which it reacts,.The chemical reactivity of a substance can refer to the variety of circumstances (conditions that include temperature, pressure, presence of catalysts) in which it reacts, in combination with the: interacts with two or more other reactants to form two or more products.forms new substances by addition of atoms from another reactant or reactants, and. 
The chemical reactivity of a single substance (reactant) covers its behavior in which it:
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
